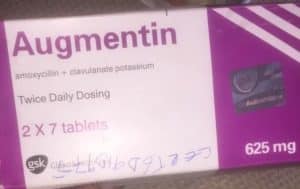
The National Agency for Food and Drug Administration Control, NAFDAC, has drawn the attention of the public to the detection of suspected falsified Augmentin 625mg Tablets in circulation within the country.
In a statement on its website, the agency said the fake drug has the following details and labelling lapses;
Product Name: Augmentin 625mg. Batch No.: 562626, Manufacturing date: April 2021, Expiry date: April 2024, and NAFDAC Reg No: 04-1928.
NAFDAC said the product failed short of the labelling requirements: “No inscription “manufactured by” is written on the label -only the address. Manufacturing and Expiry dates do not meet the acceptable format. No MAS scratch number for verification. The logo “gsk” is not properly positioned as on the original.”
It said the listed information indicates the product is falsified and counterfeited
The statement read in part, “In view of the above, NAFDAC has notified all its formations in the zones and 36 states of the federation including the FCT to carry out surveillance and mop up the falsified Augmentin tablets.
“Please note that the genuine Augmentin 625mg has legible product labelling information including date markings – expiration and manufactured dates, batch number and NAFDAC registration number.
“NAFDAC advise to wholesalers, distributors, and pharmacies is that medicines should be obtained from authorized/licensed suppliers, increased vigilance is hereby encouraged within the supply chain to avoid infiltration of the falsified product. The products’ authenticity, physical condition and labels should be carefully checked.
“NAFDAC implores healthcare providers to ensure vigilance to prevent the administration of the falsified products to unsuspecting patient. Members of the public in possession of the above suspected counterfeit product are advised to discontinue sale or use and submit stock to the nearest NAFDAC office.
“Healthcare providers and the public should notify the nearest NAFDAC office of any information concerning the distribution, sale, and use of the falsified version of the Augmentin product. Report any suspicion of adverse drug reaction and substandard and falsified medicines to NAFDAC on 0800-162-3322 or email: [email protected] “